
Advanced Needling Technique for Cosmetics
The nappage technique, otherwise known as “multi-puncture” or “micro-papular” technique, is an innovative needling method breaking into the field cosmetic dermatology. Using this skill along with Russell Health’s patent-pending procedure guides for Stem Cell Recruitment Therapy can be used to treat lack of elasticity, dry, dehydrated, aging skin and can help to improve fine lines and…
Keep Reading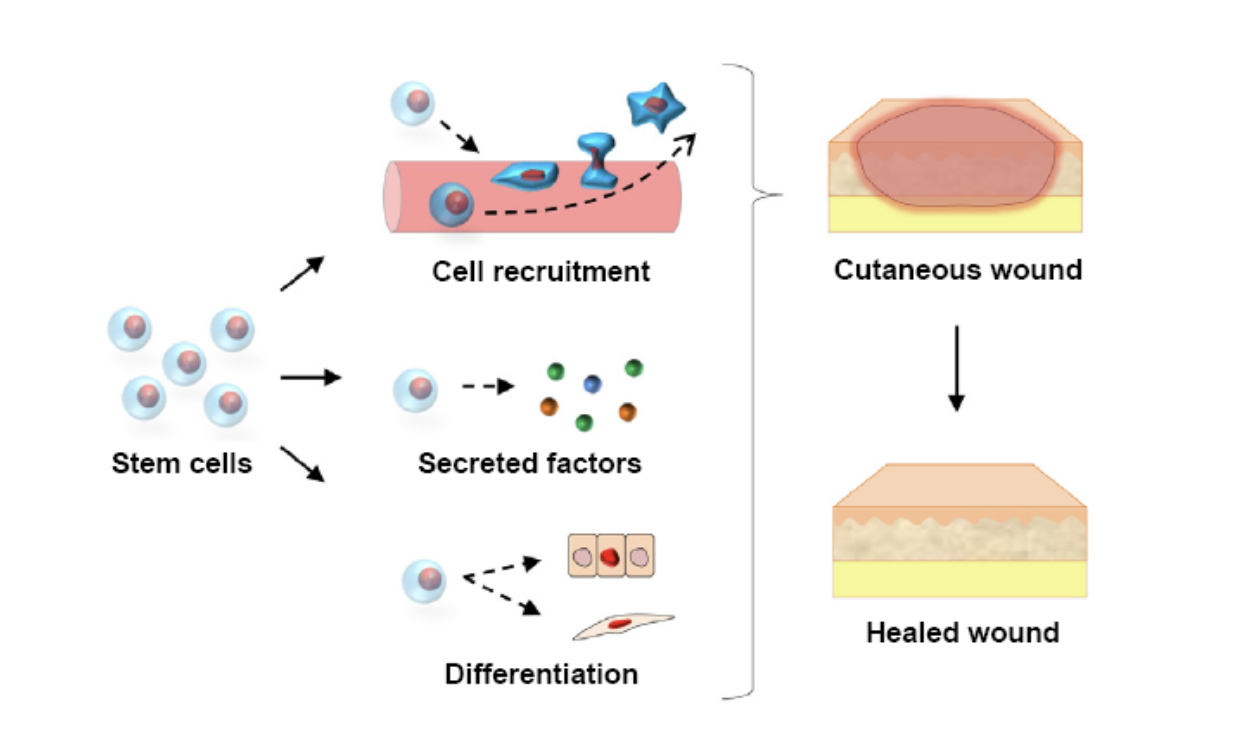
What is Regenerative Healing?
What is regenerative healing? Tissue Engineering Tissue repair and regeneration involve cell proliferation as well as the selective recruitment of circulating stem and progenitor cell populations through complex cell signaling. Thanks to modern technology, the evolution of tissue engineering was born. "Tissue engineering was developed from biomaterials and refers to the practice of combining scaffolds,…
Keep Reading